|
There are two types of light-water reactors operating in America. This means they use normal water as both a coolant and neutron moderator. Types of Light-water Reactors in the United StatesĪll commercial nuclear reactors in the United States are light-water reactors. The heat created by fission turns the water into steam, which spins a turbine to produce carbon-free electricity. The moderator helps slow down the neutrons produced by fission to sustain the chain reaction.Ĭontrol rods can then be inserted into the reactor core to reduce the reaction rate or withdrawn to increase it. Inside the reactor vessel, the fuel rods are immersed in water which acts as both a coolant and moderator. A reactor core is typically made up of a couple hundred assemblies, depending on power level. electricity, including the share generated by. Typically, more than 200 of these rods are bundled together to form a fuel assembly. Nuclear power provided 22.5 of the electricity generated by utilities in 1995 it provided about 20 of all U.S. The uranium is processed into small ceramic pellets and stacked together into sealed metal tubes called fuel rods. Radioactive decay of both fission products and transuranic. 
Three types of moderators are used at the MIT reactor: (1) ordinary or "light" water that is also used to cool the reactor core, (2) deuterated or heavy water (D 20), and (3) high-purity graphite, both of which are excellent at slowing neutrons without absorbing them.Reactors use uranium for nuclear fuel. Nuclear reactors work by containing and controlling the physical process of nuclear fission. Only a few nuclei are known to undergo fission. Since U-235 nuclei do not readily absorb the high energy neutrons that are emitted during fission, it is necessary to slow the neutrons down with a "moderator". In nuclear fission a large nucleus is split into two medium-sized nuclei. Each time a U-235 nucleus splits, it releases two or three neutrons. Uranium is the most widely used fuel for nuclear fission. Fission releases heat energy that can generate steam, which is used to spin a turbine to produce electricity1. This process is known as fission (see diagram below). Nuclear fission is a process whereby energy is released by the splitting of uranium atoms. 
When a U-235 nucleus absorbs an extra neutron, it quickly breaks into two parts. In the MIT reactor, one other group of components is essential to the maintaining and controlling a chain reaction. The arrangement of particles within uranium-235 is somewhat unstable and the nucleus can disintegrate if it is excited by an outside source. As fewer and fewer neutrons are absorbed, more and more neutrons are available to cause the splitting of uranium nuclei, until finally enough neutrons are available to sustain a chain reaction. To put the reactor into operation, the control blades are raised very slowly. 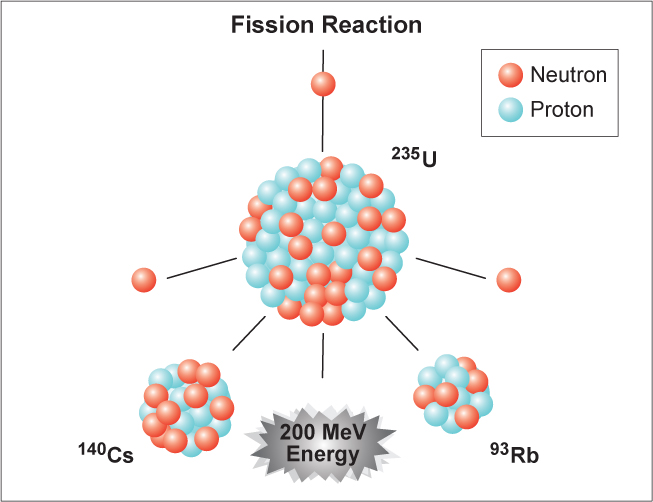
When the control blades are fully inserted, they absorb so many neutrons from the uranium that there are not enough to allow a chain reaction to continue. Nuclear power is the second-largest source of low-carbon electricity today, with 452 operating reactors providing 2700 TWh of electricity in 2018, or 10 of. When an atom splits into two parts, either through natural decay or when instigated within a lab, it releases energy. Nuclear fission leaves behind radioactive waste and. Boron has the property of absorbing neutrons without re-emitting any. Nuclear fission plants generated about 18 of electricity in the U.S. The rate of fissions in the uranium nuclei in the MIT reactor is controlled chiefly by six control blades of boron-stainless steel which are inserted vertically alongside the fuel elements. When it is in operation, the central active core contains a huge number of neutrons traveling in every direction at very high speeds. The MIT Research Reactor is used primarily for the production of neutrons. Hence, the possibility exists for creating a chain reaction. This process is known as fission (see diagram below). This energy is carbon-free, meaning that like wind and solar, it does not directly produce carbon dioxide (CO 2) or other greenhouse gases that contribute to climate change. When a U-235 nucleus absorbs an extra neutron, it quickly breaks into two parts. Nuclear energy is energy made by breaking the bonds that hold particles together inside an atom, a process called nuclear fission. The arrangement of particles within uranium-235 is somewhat unstable and the nucleus can disintegrate if it is excited by an outside source. 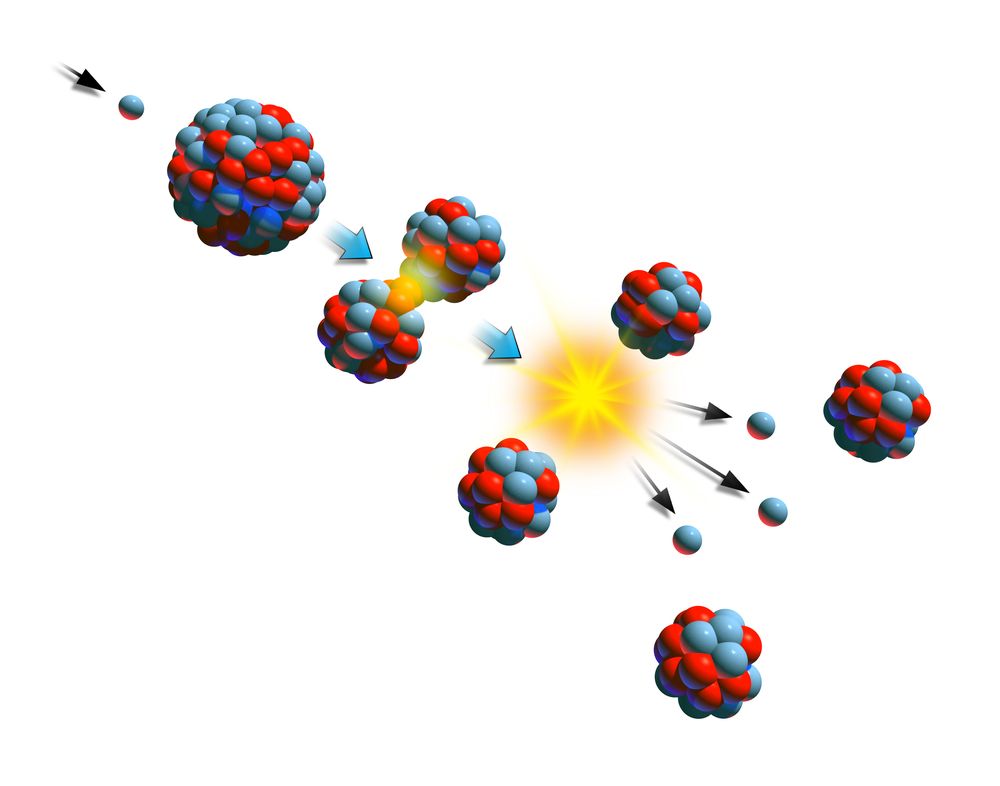
In the nucleus of each atom of uranium-235 (U-235) are 92 protons and 143 neutrons, for a total of 235. The amount of energy released during nuclear fission is millions of times more efficient per mass than that of coal considering only 0.1 percent of the original nuclei is converted to energy.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
